 
The system is intended to be used with leads and associated extensions that are compatible with the system. 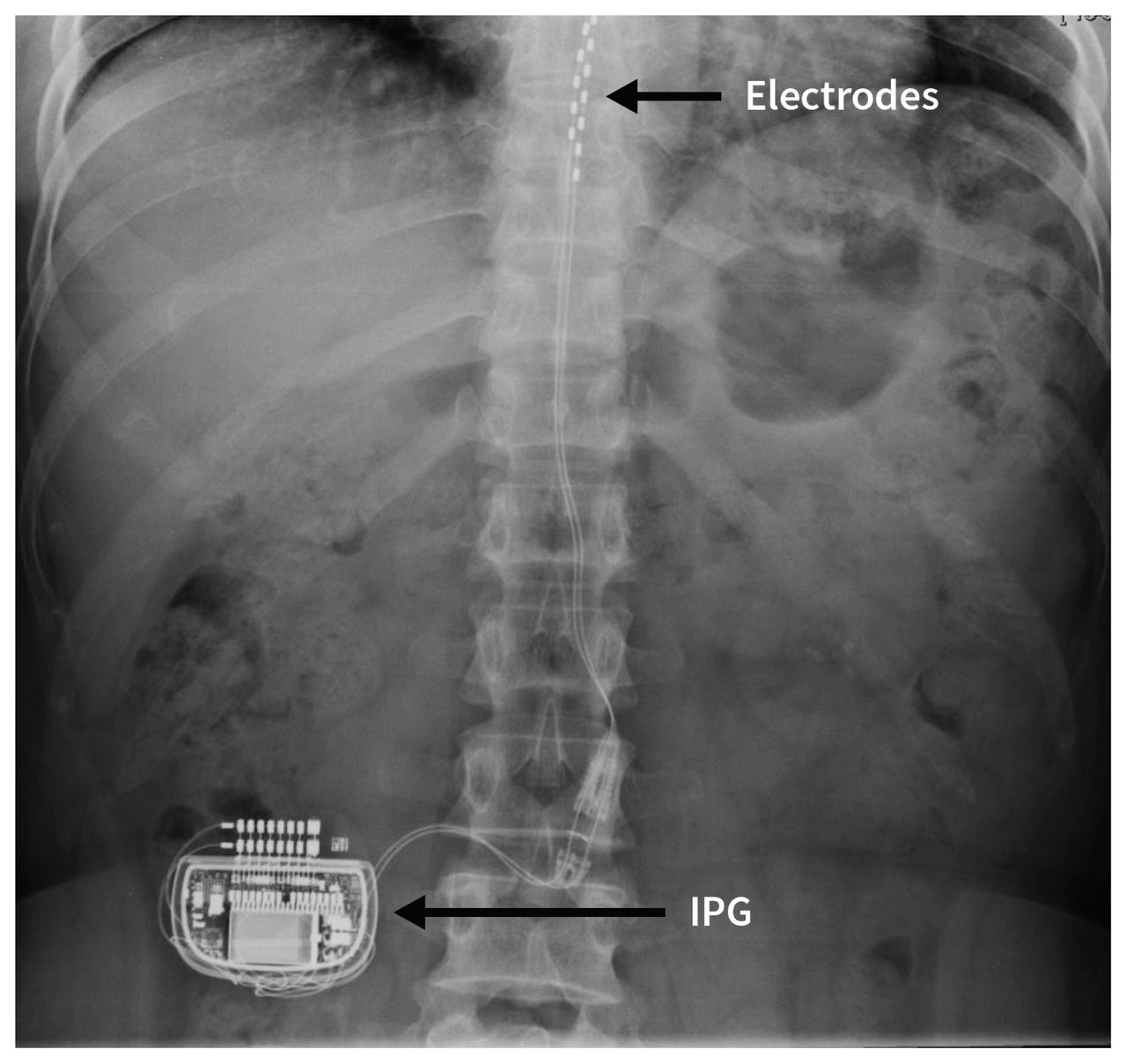
This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. Alere discontinued selling its INRatio blood coagulation monitors in 2016.Spinal Column Stimulation (SCS) Systems Intended Use Jude Medical cases took place before Abbott acquired them in 2017. 
In company statements provided to MassDevice, Abbott noted that the matters involved in both the Alere and St. Within months, a Class I–level recall was underway, according to the government. 
In August 2016, the company disclosed to FDA there had been 729 battery depletion events, with two deaths and 29 instances of loss of pacing. In fact, the company was aware of two reported serious injuries and one death, the government claims. Jude Medical failed to disclose premature battery depletion problems involving its Fortify, Fortify Assura, Quadra and Unify devices - even though the company was aware as early as 2013 that lithium clusters formed on device batteries were causing the batteries to short.īy late 2014, St Jude Medical asked FDA to approve a change to prevent lithium clusters from draining batteries, saying at the time that there were no serious injuries or deaths, according to the DOJ. Jude knowingly caused the submission of false claims and failed to inform the FDA with critical information about prior injuries and a death which, had the FDA been made aware, would have led to a recall,” Lenzner said in a DOJ release posted yesterday. government will not pay for devices that are unsafe and risk injury or death,” said acting U.S. “Medical device manufacturers have an obligation to be truthful with the Food and Drug Administration (FDA), and the U.S. Jude Medical for $25 billion in January 2017. Jude Medical knowingly sold ICDs with battery problems between November 2014 and October 2016. District Court in Maryland claimed that St. Department of Justice news release posted yesterday. “The government expects companies to be proactive in investigating issues affecting patient safety,” Honig said in a U.S. “Health care companies have an obligation to be candid and clear in their disclosures to the FDA,” said acting U.S. The government claims that the company didn’t take appropriate actions to correct the problems until a Class I-level recall in 2016. Blood coagulation monitoring is essential to determine safe dosages.Īlere allegedly knew that the software algorithm used in each version of its INRatio monitors contained a material defect. District Court in Newark claimed that from 2008 to 2016, Alere knowingly sold defective INRatio blood coagulation monitors used by people taking anticoagulant drugs, such as warfarin. Jude Medical lawsuit in federal court in Maryland. Without admitting liability in either case, Abbott will pay $38.75 to settle the Alere-related lawsuit in federal court in Newark, N.J., and $27 million to resolve the St. The Abbott Park, Ill.-based medtech giant will pay nearly $66 million as part of the settlements. Jude Medical and Alere - two companies it spent billions of dollars to acquire in 2017. Department of Justice over False Claims Act lawsuits involving St. Abbott (NYSE:ABT) has reached settlements with the U.S. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
